Aluminum brazing is a process that involves joining two pieces of aluminum together using a filler material. One of the key components of this process is the use of aluminum brazing flux. This flux is a chemical compound that is applied to the surface of the aluminum prior to brazing.
The primary function of aluminum brazing flux is to remove any oxide layers that may be present on the surface of the aluminum. These oxide layers can interfere with the brazing process and prevent a strong joint from forming. The flux works by reacting with the oxide layer and dissolving it, allowing the filler material to bond more effectively with the aluminum.
There are several different types of aluminum brazing flux available, each with its own advantages and disadvantages. Corrosive flux, for example, has been the standard for many years and is effective at removing oxide layers. However, it can also be corrosive to the aluminum and may require additional cleaning after brazing. Non-corrosive flux, on the other hand, is less likely to damage the aluminum but may not be as effective at removing oxide layers. By understanding the different types of aluminum brazing flux available, it is possible to choose the best one for a particular application.
What is Aluminum Brazing Flux?
Aluminum brazing flux is a material used in the process of aluminum brazing, which involves joining two or more pieces of aluminum or aluminum alloys together using a filler metal. The flux is applied to the surfaces of the aluminum parts to be joined before the brazing process begins.
The main function of the aluminum brazing flux is to remove any oxide layer that may have formed on the surface of the aluminum parts. The oxide layer can prevent the filler metal from bonding properly with the aluminum, leading to weak joints. The flux also helps to wet the surface of the aluminum, allowing the filler metal to flow evenly over the joint.
There are two main types of aluminum brazing flux: corrosive and non-corrosive. Corrosive flux contains chloride and fluoride salts and is water-soluble. The residues left after the brazing process must be washed off the parts with a solution of nitric acid and water. Non-corrosive flux, on the other hand, is not water-soluble and does not require washing after brazing.
The choice of flux depends on the specific application and the materials being joined. The brazing temperature, the type of aluminum alloys being used, and the filler metal also affect the choice of flux. Torch brazing is a common method of aluminum brazing, but induction brazing is also used in some applications.
In summary, aluminum brazing flux is a critical component of the aluminum brazing process. It helps to remove oxide layers, promotes wetting, and ensures strong joints between aluminum parts. The choice of flux depends on the application and materials being joined, as well as the brazing method and temperature.
Types of Aluminum Brazing Fluxes
When it comes to aluminum brazing, there are several types of fluxes available on the market. These fluxes are designed to clean the surface of the aluminum and promote the wetting and flow of the filler material, resulting in strong and reliable joints.
Corrosive Fluxes
Corrosive fluxes are typically made of a mixture of hydrochloric and hydrofluoric acids. These fluxes are highly effective at removing oxides and other contaminants from the surface of the aluminum, but they can also be highly corrosive and damaging to the aluminum if not used properly. As a result, corrosive fluxes are not recommended for use on aluminum alloys that are prone to corrosion, such as the 2000 and 7000 series.
Non-Corrosive Fluxes
Non-corrosive fluxes are designed to be less aggressive than corrosive fluxes, making them a better option for use on aluminum alloys that are prone to corrosion. These fluxes are typically made of potassium or sodium compounds and are effective at removing oxides and other contaminants from the surface of the aluminum.
Flux-Cored
Flux-cored aluminum brazing filler metals are designed to be used with a flux core, which eliminates the need for a separate fluxing step. These filler metals are typically made of aluminum-silicon alloys and are effective at brazing a wide range of aluminum alloys.
Dispensable Fluxes
Dispensable fluxes are pre-applied to the filler metal in a controlled environment, which ensures that the flux is evenly distributed and reduces the risk of contamination. These fluxes are typically used in automated brazing processes and are effective at brazing a wide range of aluminum alloys.
Nocolok® Flux
Nocolok® flux is a non-corrosive flux that is an industry standard for aluminum brazing. It is effective at brazing all 1000 and 3000 series aluminum alloys and is often used for aluminum heat exchangers and tube-to-tube joints.
KX Type Flux
KX Type flux is similar to Nocolok® flux but contains a small amount of cesium. This flux has similar melting characteristics to 100% Nocolok® flux and is effective at brazing all 1000 and 3000 series aluminum alloys.
CX Type Flux
CX Type flux is a non-corrosive flux that is specifically designed for use on aluminum alloys that are prone to corrosion, such as the 2000 and 7000 series. This flux is effective at removing oxides and other contaminants from the surface of the aluminum and promoting the wetting and flow of the filler material.
Solvay Fluor
Solvay Fluor is a non-corrosive flux that is specifically designed for use on aluminum alloys that are prone to corrosion. This flux is effective at removing oxides and other contaminants from the surface of the aluminum and promoting the wetting and flow of the filler material.
Aluminum Flux
Aluminum flux is a corrosive flux that is designed for use on aluminum alloys that are not prone to corrosion. This flux is highly effective at removing oxides and other contaminants from the surface of the aluminum, but it can also be highly corrosive and damaging to the aluminum if not used properly.
In summary, there are several types of aluminum brazing fluxes available on the market, each with its own unique properties and benefits. It is important to choose the right flux for the job, taking into account the type of aluminum alloy being brazed and the brazing process being used.
Choosing the Right Aluminum Brazing Flux
When it comes to aluminum brazing, choosing the right flux is crucial to ensure a strong and reliable joint. Here are some factors to consider when selecting an aluminum brazing flux:
Base Alloy
The first step in selecting a brazing flux is to consider the base alloy being used. For instance, if you are brazing aluminum, you will need an aluminum brazing flux. Superior Flux & Mfg. Co. recommends their No. 20 flux powder for open-air brazing of aluminum with AL718 brazing wire.
Flux Type
There are several types of aluminum brazing fluxes available, including flux-cored, dispensable flux, Nocolok® flux, KX type flux, CX type flux, and Solvay fluor. Each type has its own unique properties and benefits, so it’s important to choose the one that best suits your specific application.
Melting Temperature
Another important factor to consider is the melting temperature of the flux. The active temperature range of the flux should be compatible with the melting temperature of the base metal being brazed. For example, if the base metal melts at 600°C, the flux should have an active temperature range above 600°C.
Heating Methods
Different heating methods may require different types of flux. For example, if you are using a torch to heat the joint, you may need a flux that can withstand the high temperatures produced by the torch. On the other hand, if you are using an induction heating method, you may need a flux that is more reactive to the heat.
Oxides
Fluxes are designed to dissolve surface oxides and protect the cleaned surfaces from re-oxidation. As such, it’s important to choose a flux that is effective at removing oxides and preventing re-oxidation.
In conclusion, selecting the right aluminum brazing flux is critical to achieving a strong and reliable joint. By considering factors such as base alloy, flux type, melting temperature, heating methods, and oxides, you can choose a flux that will meet the specific requirements of your application.
Aluminum Brazing Process
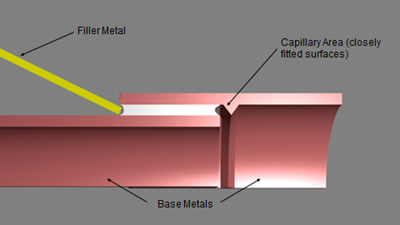
Aluminum brazing is a joining process used to bond two or more aluminum base metals together using a filler metal and an aluminum brazing flux. The process is commonly used in manufacturing heat exchangers, automotive components, and other industrial applications.
Preparation
Before brazing, the base metals must be cleaned and degreased to remove any surface contaminants that could interfere with the brazing process. The filler metal is then selected based on the application and specification requirements. The aluminum brazing flux is also selected based on the brazing process and the type of base metals used.
Brazing
The brazing process involves heating the base metals and the filler metal to a temperature above the melting point of the filler metal but below the melting point of the base metals. The aluminum brazing flux is then applied to the joint area to promote wetting and bonding of the filler metal to the base metals.
Furnace brazing is commonly used for high-volume manufacturing, while torch brazing is used for smaller production runs and repairs. The brazing process must be carefully controlled to prevent overheating, which can result in weakened joints and leaks.
Post-Braze Washing
After brazing, the joint must be cleaned to remove any residual flux or oxides that may have formed during the brazing process. Post-braze washing is typically done using a mild alkaline solution or a combination of hot water and detergent.
Inspection
Inspection personnel must visually inspect the braze joints to ensure that they meet the required specifications. The visual appearance of the joint is an important indicator of the quality of the bond. The joint should be smooth, free of voids, and have good wetting of the filler metal to the base metals.
Aluminum brazing provides a strong bond with good strength and corrosion resistance. The use of non-corrosive fluxes is becoming more common in the automotive industry due to their insulator properties and the elimination of flux migration. Aluminum brazing flux is available in paste forms and as cesium compounds for connecting block type joints.